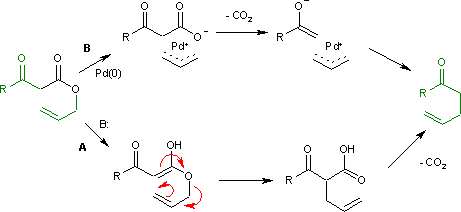
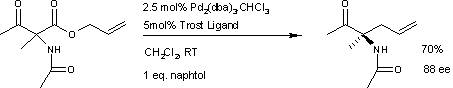Palladium/N-heterocyclic carbene catalysed regio and diastereoselective reaction of ketones with allyl reagents via inner-sphere mechanism | Nature Communications
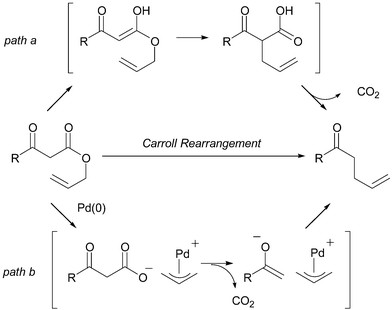
Asymmetric Carroll rearrangement of allyl α-acetamido-β-ketocarboxylates catalysed by a chiral palladium complex - Chemical Communications (RSC Publishing) DOI:10.1039/B505105C

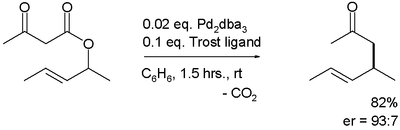
Enantioselective Palladium‐Catalyzed Decarboxylative Allylic Alkylations - You - 2006 - Angewandte Chemie International Edition - Wiley Online Library
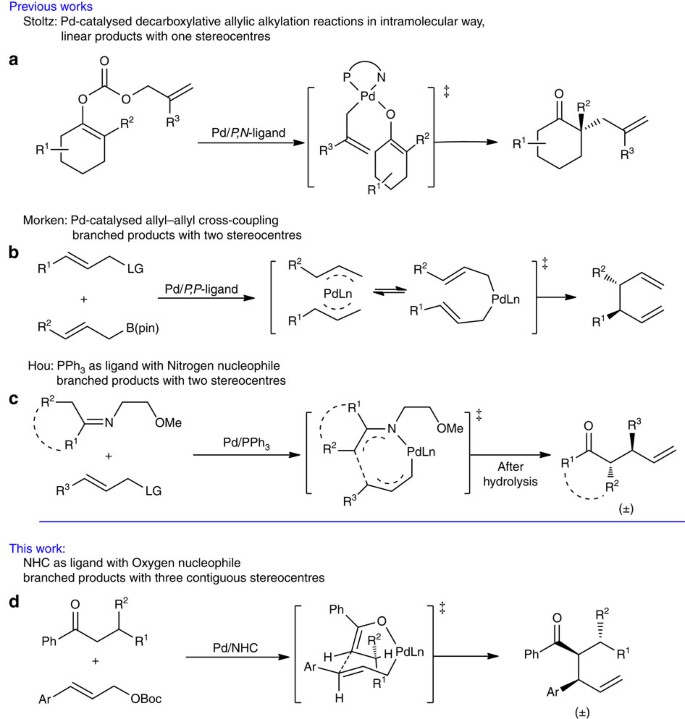
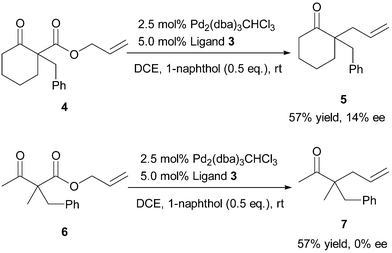
Palladium/N-heterocyclic carbene catalysed regio and diastereoselective reaction of ketones with allyl reagents via inner-sphere mechanism | Nature Communications

Palladium‐Catalyzed Decarboxylative Asymmetric Allylic Alkylation: Development, Mechanistic Understanding and Recent Advances - James - 2019 - Advanced Synthesis & Catalysis - Wiley Online Library

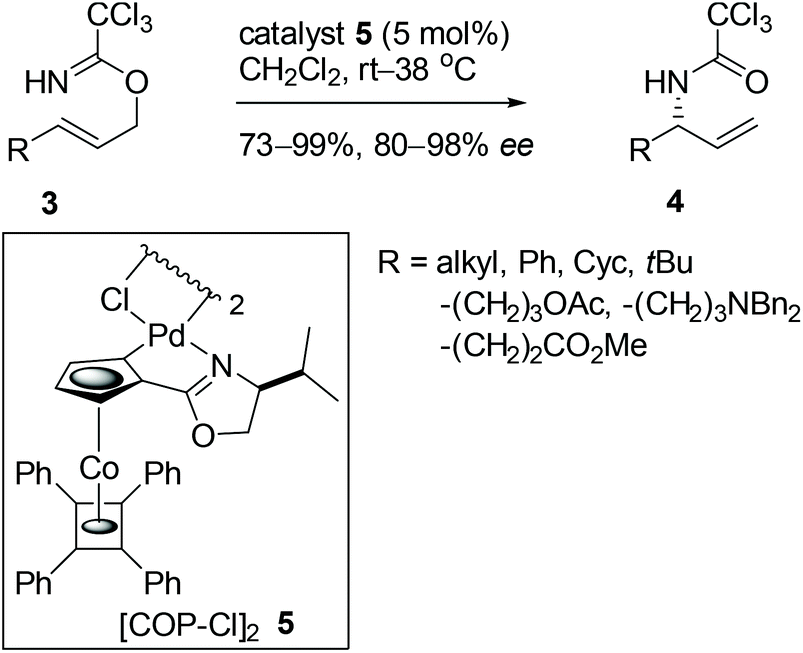
CpRuII-chiral bisamidine complex catalyzed asymmetric Carroll-type decarboxylative allylation of β-keto allyl esters - ScienceDirect
Asymmetric Carroll rearrangement of allyl α-acetamido-β-ketocarboxylates catalysed by a chiral palladium complex - Chemical Communications (RSC Publishing) DOI:10.1039/B505105C

Palladium‐Catalyzed Decarboxylative Asymmetric Allylic Alkylation: Development, Mechanistic Understanding and Recent Advances - James - 2019 - Advanced Synthesis & Catalysis - Wiley Online Library